Activated Carbons are the most powerful
adsorbents known. It is basically a solid material consisting mainly of pure
carbon. A characteristic feature is its porous structure and the resulting
immense surface area which may be as large as 1500 m2/gm. Due to its exceptional
adsorption qualities, activated carbon is widely used in process destined to
purify, discolor, recuperate and remove odors at low cost and superior
efficiency. Activated carbons work on the principle of adorption. Adsorption is
an interfacial process involving the collection of gaseous or solute components
on the surface of adsorbent solids. This phenomenon is associated with physical
attractive forces that bind gaseous and solute molecules commonly known as
Van-der-Waals forces. Adsorption is thus a physical process, i.e. the
substances adsorbed on the solid do not undergo any chemical reaction with the
latter. The adsorbing solid is referred to as adsorbent and the substance to be
adsorbed from the liquid or the gas phase as the solute.
|
The adsorption power and rate is determined by the kind
of activated carbon, the particle size, the pore size and its distribution.
|
|
|
|
|
|
|
|
Pore Sizes and its distribution
|
|
|
|
When
the carbon is activated it leads to opening of various pores in its
structure.
|
|
|
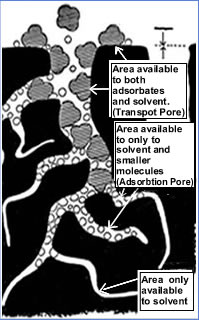
The
pores with diameters exceeding 500 A° are called macropores or
the transport pores. These pores are larger pores and are not
responsible for adsorption directly but they act as wide paths through which
organic molecules penetrate. Thus they transport the adsorbate to the
adsorption site, which are located inside the particle. A full network of
transport pores needs to be distributed through out the carbon particle to
take the adsorbate to the smallest adsorption pore site.
|
|
|
Pores
with diameter below 50 A° are called micropores or the adsorption pores.
These are the finest pores , which are located inside the carbon particle
where the adsorption actually takes place.
|
|
|
The
most common parameter to check the adsorption power of activated carbon is
called methylene blue adsorption. The methylene blue indicator is a chemical
having particle size 12 A° which prevents it from adsorbing on the adsorption
pores, which are smaller than 12 A°. That is why an activated carbon with a
uniform and better pore size distribution will not give high methylene blue
value but will work better as it will adsorb impurities which are very small
in size.
|
|
|
For
activated carbon to give good performance it needs to have both these pores
in good proportion | | | | | | | | | | | | | | | | | | |
|
|
|
|
|
|
|
|
|
|
|
|


